JAN SCHMORANZER USES MODERN MICROSCOPES TO GET A CLOSER LOOK AT THE BUILDING BLOCKS OF HUMAN CELLS AND HELP TO DEVELOP NEW DIAGNOSIS TECHNIQUES.
In a laboratory at Berlin’s Charité university hospital, Jan Schmoranzer, 39, takes a plastic slipcase with 96 compartments out of a cabinet: “There are cancer cells in here,” he says, placing the slipcase in a black container under the lens of his microscope. It’s the BD Pathway 855 optical microscope, one of the most modern devices on the market and one that has little in common with the tabletop microscopes we remember from our schooldays. At the push of a button a little robotic arm swings past the lens, grasps the cell-filled container and aligns it. Next to it on the right, a big computer screen takes pride of place, displaying the close-up images of the cell culture.
“The cells come from a woman who died of cervical cancer in Baltimore in 1951,” says the scientist. They established a cell line that was cultured and passed from lab to lab around the globe—and still is. Scientists have been using it for a wide range of experiments for decades—including those at Charité Berlin, one of Europe’s biggest university hospitals, where Schmoranzer, a physicist, works as head of the Microscopy Department.
Schmoranzer is an expert in the microscopy of cell samples and, as such, is one of a handful of young scientists who are in demand the world over. That’s because modern medicine now relies more and more on imaging techniques in the course of its research. Such techniques enable scientists to look right into the heart of cells. This close-up view delivers valuable knowledge about the chemistry of the cells: A better understanding of the metabolic processes in the cells will ideally give scientists knowledge of what’s going wrong in the body when disease strikes, thus enabling them to develop targeted medication.
However, by no means can everyone operate these modern microscopes. They are too complex in the way they work, too varied in their range of applications. And so a new profession has grown up in recent years, one which is now in urgent demand in the labs of the world: the bioimaging expert. “Many institutes or large laboratories are currently in the process of establishing their own departments with experts like me,” says Schmoranzer. Charité Berlin enticed Schmor anzer away from New York, where he had spent 13 years working for Rockefeller and Columbia Universities.
He moves the BD Pathway 855 optical microscope a little to the right with the joystick. The images are sent from the lens to a digital camera, and from there they are transmitted to the computer screen. This microscope, which sells for 350,000 euros, allows the scientist to get right up close to the cells, as close as 200 nanometers, visualizing the smallest components of the cells with a staggering degree of magnification. It’s a real microcosm that Schmoranzer is working on. By way of comparison, the diameter of a human hair, at 7,500 nanometers, is 350 times larger than many of the cell components Schmoranzer is visualizing.
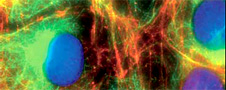
Suddenly the screen is filled with white dots in a gray, shapeless form. “We’re in the cell nucleus,” explains Schmoranzer. “The gray haze around the dots is the rest of the cell space.” If a cell contains white dots, it’s diseased; healthy cells have fuzzy gray dots. Why, no one knows — yet. But even the first vague findings provide a measure of help.
THE FIGHT AGAINST LEUKEMIA
Schmoranzer and his fellow scientists at Charité are hot on the trail of leukemia. And the Pathway is going to help them by using clever technology to conduct mass experiments: thousands of diseased white blood cells will be mixed with various chemicals and put under the microscope. The computer will then automatically distinguish healthy blood cells from diseased ones, thereby simplifying the painstaking process of drug discovery. “We want to find a solution that either genetically modifies diseased cells and reprograms them into healthy ones, or kills them off,” says the physicist. He is currently writing a piece of software to handle the automatic image recognition—and practicing on the cancer cells from Baltimore.
Now Schmoranzer wants to get even closer. He unscrews the lens offering 20 times magnification and replaces it with one that magnifies images 40 times. The digital camera does the rest. “It’s only a 1.4-megapixel camera, but its pixels are a lot smaller and more sensitive to light than those of conventional cameras,” says Schmoranzer. It therefore makes even the smallest of structures visible.
Yet even that doesn’t quite do the trick. At this resolution, the cells appear so endlessly big that you need to locate to the precise segment you want to look at. Schmoranzer employs a complex technique to find it: the Pathway is what’s known as a fluorescence microscope. With the help of molecular-biological methods it can make precise sections of the cell light up.
In the case of the cancer cells under his Pathway, Schmoranzer is trying to find his way to a protein in the cell nucleus. In a bid to make it visible, the scientist delves into the animal world’s bag of tricks—the secrets of a jellyfish, to be more precise. Jellyfish have a special protein that glows green at a certain incidence of light. Special enzymes can be used to combine the DNA strings of both mole – cules—thereby making the cell produce a double protein instead of its normal protein; the double protein turns green when exposed to a beam of light. This is where Schmoranzer’s own special brand of expertise comes into play: Using the microscope’s many different filters and light sources he figures out a clever way of lighting the sample so that the protein is illuminated at just the right angle and just the right wavelength. It’s only through his work that the protein starts to fluoresce—and becomes visible to the observer.
THE BEAUTY OF THE CELLS
He’s in a hurry because it’s his final week working here—he’s about to move to the Free University of Berlin (FU Berlin). There he will build a microscope capable of getting to within 20 nanometers of the cell samples, in other words ten times more powerful than the microscope at Charité. There are very few microscopes of such high resolution around. Most of them are in the US, which is where Schmoranzer himself first came into contact with them: on an internship at New York’s Rockefeller University, which he embarked upon after completing his physics degree at Aachen. Looking through the eyepiece for the very first time, he knew that was it for him: “Each cell is as individual as a person,” he raves. “In New York I saw the beauty of their arrangement, I got a sense of the extent of their impact on life.” The young German’s enthusiasm won the professors over. They invited him to pursue his Ph.D. with them. His subject was the high-resolution imaging of protein transportation through the outer skin of the living cell.
This specialization was quite an obvious choice for Schmoranzer, being a keen photographer. His father gave him his first single-lens reflex camera at 15, after which he spent countless evenings on the balcony trying to capture the perfect sunset. Later, Schmoranzer even had his own photography exhibitions. He has already won prizes for his fascinating cell photographs.
SECRETS OF THE CELL WALL
His new job at FU Berlin may well bring him other prizeworthy photos. “I want to get even closer to the cell surface at FU Berlin,” says Schmoranzer as he pulls off his disposable gloves. Unlike his job at Charité, Schmoranzer will not be writing computer programs here. He will be helping to visualize cell mechanisms that are, as yet, virtually unknown. The spatial distribution and interaction of the molecules in the outer membrane are what he plans to study in more detail. By this he means the chemical compounds that act like switches to start or stop the cells’ actions at the outer cell walls. Here too, Schmoranzer will experiment with mirrors, lenses and fiber optic cables until he finds just the right settings to make the structures on the membrane visible. If he succeeds, scientists could discover how commands are issued and followed at the outer cell wall. This could ultimately give man the power of control over cells.
But for now Schmoranzer flicks the switch on the computer. That’s enough for today. The cells, flickering as big as a hand on the screen until barely a moment ago, vanish into nothingness. Suddenly the closeness appears remote indeed.
(Text: Jan Rübel, Photos: Paul Hahn, Jan Schmoranzer)
Bilfinger Berger Magazine 2/2009